Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Okazaki, Hiro; Shibano, Koya; Abe, Katsuo; Sumi, Mika; Kayano, Masashi; Kageyama, Tomio; Mason, P.*
Proceedings of INMM 57th Annual Meeting (Internet), 7 Pages, 2016/07
The reference materials used for accountancy analysis by IDMS, referred to as LSD spikes allows to obtain accurate measurement results for a variety of nuclear materials under potentially difficult handling or measurement conditions. Because of expected difficulties in a stably long term supply of plutonium reference materials, which are the main source of plutonium for LSD spikes preparation, the JAEA's PFDC decided to evaluate the possibility of using domestically available MOX powders as a plutonium source material for LSD spikes. Therefore, PFDC started to separate, purify and characterize plutonium in MOX powders for plutonium reference materials (MOX-Pu) preparation that are suitable for source of LSD spikes in cooperation with the US DOE's NBL. The detailed preparation procedure of the latest MOX-Pu standard material and results of intercomparison for verification are presented.
 -factors for k
-factors for k -based neutron-induced prompt
-based neutron-induced prompt  -ray analysis
-ray analysisMatsue, Hideaki; Yonezawa, Chushiro
Journal of Radioanalytical and Nuclear Chemistry, 255(1), p.125 - 129, 2003/01
Times Cited Count:8 Percentile:49.89(Chemistry, Analytical)no abstracts in English
 -ray analysis using k
-ray analysis using k standardization
standardizationMatsue, Hideaki; Yonezawa, Chushiro
Journal of Radioanalytical and Nuclear Chemistry, 249(1), p.11 - 14, 2001/06
no abstracts in English
 2000/12)
2000/12)Kobayashi, Hideo; ; ; ; ; *; *
JNC TN8440 2001-005, 33 Pages, 2001/02
Four mass spectrometers are used for plutonium and uranium isotopic composition analysis in the Plutonium Fuel Center. Analytical errors of the analysis was evaluated based on routine control data obtained with measurement of reference materials. It was confirmed that the errors satisfied the International Target Values for safeguard analysis, and the random error of the analysis was improved by changing conventional method to total evaporation method.
Yonezawa, Chushiro
Analytical Sciences, 12, p.605 - 613, 1996/08
Times Cited Count:27 Percentile:71.55(Chemistry, Analytical)no abstracts in English
Yonezawa, Chushiro; ; *; Sasajima, Fumio; R.P.Putra*
2nd Int. k Users Workshop Proceedings, 0, p.129 - 133, 1996/00
Users Workshop Proceedings, 0, p.129 - 133, 1996/00
no abstracts in English
Yonezawa, Chushiro; *
Bunseki Kagaku, 45(5), p.435 - 440, 1996/00
no abstracts in English
 -ray analysis of boron with cold and thermal neutron guided beams
-ray analysis of boron with cold and thermal neutron guided beamsYonezawa, Chushiro; A.K.H.Wood*
Analytical Chemistry, 67(24), p.4466 - 4470, 1995/12
Times Cited Count:35 Percentile:79.79(Chemistry, Analytical)no abstracts in English
Yonezawa, Chushiro; ; Hoshi, Michio; Ito, Yasuo*
Proc. of Int. Trace Analysis Symp. 94; ITAS 94, 0, p.81 - 84, 1994/00
no abstracts in English
 December 1988
December 1988JAERI-M 89-058, 342 Pages, 1989/05
no abstracts in English
Shibano, Koya; Okazaki, Hiro; Sumi, Mika; Kayano, Masashi; Matsuyama, Kazutomi; Kageyama, Tomio; Fujiwara, Hideki*; Furuta, Koya*; Yamaguchi, Kazuya*; Saito, Shingo*
no journal, ,
no abstracts in English
Miyajima, Yusuke*; Saito, Ayaka*; Kagi, Hiroyuki*; Yokoyama, Tatsunori; Hirata, Takafumi*
no journal, ,
no abstracts in English
Miyajima, Yusuke*; Saito, Ayaka*; Kagi, Hiroyuki*; Yokoyama, Tatsunori; Hirata, Takafumi*
no journal, ,
Calcium carbonates are ubiquitously present throughout the Earth history as animal shells, speleothems, fault-related vein fillings, and hydrothermal or cold-seep precipitates. Direct dating of carbonates provides valuable information on paleoenvironmental change, tectonics, and fluid and material cycling. U-Pb dating using high spatial-resolution LA-ICP-MS is a key technique to date natural carbonates. In situ U-Pb dating by LA-ICP-MS needs matrix-matched reference materials to correct matrix-dependent elemental fractionation in LA-ICP-MS. Roberts et al. (2017) demonstrated that a natural calcite cement WC-1 is suitable as a calcite reference material. However, the WC-1 calcite has an inhomogeneous distribution of U and Pb and lacks concordance in the U-Pb system. In this study, we synthesized novel calcite reference materials with homogeneous U and Pb concentrations and isotope ratios. We incorporated U and Pb into calcite through heat-induced crystallization from U, Pb-doped amorphous calcium carbonate (ACC). The homogeneity of the U/Ca and Pb/Ca ratios in the synthetic calcite was generally better than 8% and 13%, respectively, in relative standard deviation. The 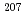 Pb/
Pb/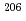 Pb ratio of the synthetic calcite was homogeneous within ~1% errors, whereas the
Pb ratio of the synthetic calcite was homogeneous within ~1% errors, whereas the  U/
U/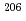 Pb ratio was less homogeneous (3%-11% errors). To test the usability of the synthetic calcite, we dated WC-1 using the synthetic calcite for correction of elemental fractionation. We calculated the nominal
Pb ratio was less homogeneous (3%-11% errors). To test the usability of the synthetic calcite, we dated WC-1 using the synthetic calcite for correction of elemental fractionation. We calculated the nominal  U/
U/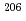 Pb ratio in the synthetic calcite from its U and Pb concentrations. We then obtained the fractionation factor to correct bias between the isotope ratios and the nominal value. We could accurately date WC-1 with an ~3% precision. If the isotopic compositions of the synthetic calcite are certified by isotope-dilution technique, we could date natural carbonates with
Pb ratio in the synthetic calcite from its U and Pb concentrations. We then obtained the fractionation factor to correct bias between the isotope ratios and the nominal value. We could accurately date WC-1 with an ~3% precision. If the isotopic compositions of the synthetic calcite are certified by isotope-dilution technique, we could date natural carbonates with  10% precisions using the synthetic reference calcite.
10% precisions using the synthetic reference calcite.